Photocatalytic conversion of CO2 into fuels, long constrained by low efficiency and unclear mechanisms, is being advanced by researchers at Chiba University through the isolation and quantification of the interaction between photocatalytic and photothermal effects in CO2-to-methane conversion.
Their work, published in the Journal of the American Chemical Society, reports methane production rates of up to 10 millimoles per gram of catalyst per hour using a Ru–Ni–ZrO2 system, placing the results among the highest reported for this class of reactions.
Photocatalytic CO2 reduction relies on light-induced electron excitation to drive chemical transformations, while photothermal effects involve heat generated under irradiation that accelerates reaction kinetics. Distinguishing between these mechanisms has been a persistent challenge, as both processes often occur simultaneously under experimental conditions. This ambiguity has limited the ability to optimize catalyst design for industrial-scale applications.
The Chiba University team, led by Professor Yasuo Izumi, approached this problem by systematically varying light intensity and temperature conditions. By using ultraviolet-visible irradiation ranging from 90 to 900 milliwatts per square centimeter and introducing controlled cooling, the researchers were able to separate thermal and non-thermal contributions to the reaction.
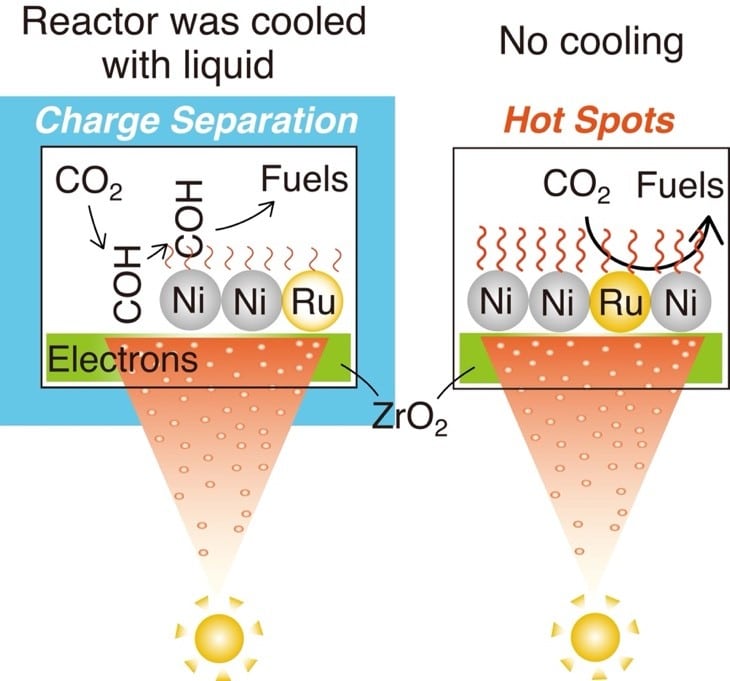
Under conditions without temperature control, the Ru–Ni–ZrO2 catalyst exhibited a methane production rate exceeding 7.9 millimoles per gram per hour, more than 2.7 times higher than a Ni–ZrO2 benchmark catalyst. In this regime, photothermal effects dominated the reaction pathway. CO2 molecules adsorbed directly onto ruthenium-nickel active sites were dissociated into carbon monoxide and oxygen with an activation energy of 0.45 electronvolts, significantly lower than the 0.79 electronvolts required for nickel-only systems.
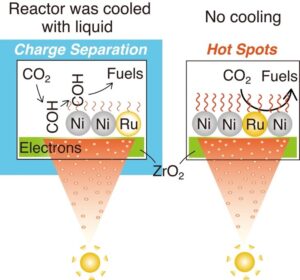
When temperature was stabilized at 295 Kelvin through a cooling bath, the reaction shifted toward a predominantly photocatalytic mechanism. In this mode, light-generated charge carriers on the zirconium dioxide surface formed intermediate OCOH species at oxygen vacancy sites. These intermediates were subsequently transferred to nickel sites for hydrogenation into methane. The presence of localized thermal hotspots on nickel, reaching temperatures up to 126 degrees Celsius under high irradiation, further enhanced reaction rates beyond what would be expected from thermal processes alone.
This dual-mechanism behavior highlights a critical design consideration for next-generation catalysts. Rather than treating photocatalytic and photothermal processes as competing effects, the study demonstrates that their interaction can be leveraged to improve overall efficiency. The relative contribution of each mechanism is determined by operating conditions such as temperature and light intensity, suggesting that reactor design and process control could play as important a role as catalyst composition.
Despite the reported efficiency gains, significant barriers remain for commercial deployment. Photocatalytic systems must achieve not only high reaction rates but also long-term stability, scalability, and integration with renewable energy sources. Additionally, the conversion of CO2 into methane, while valuable as a fuel, requires further processing infrastructure and raises questions about lifecycle emissions if the methane is ultimately combusted.
The research also points toward broader applications beyond methane synthesis. The ability to control reaction pathways could enable the production of more complex hydrocarbons and alcohols, which offer higher economic value and greater flexibility as chemical feedstocks. However, these pathways typically involve more complex reaction networks and require further advances in catalyst selectivity and stability.